|
|
|
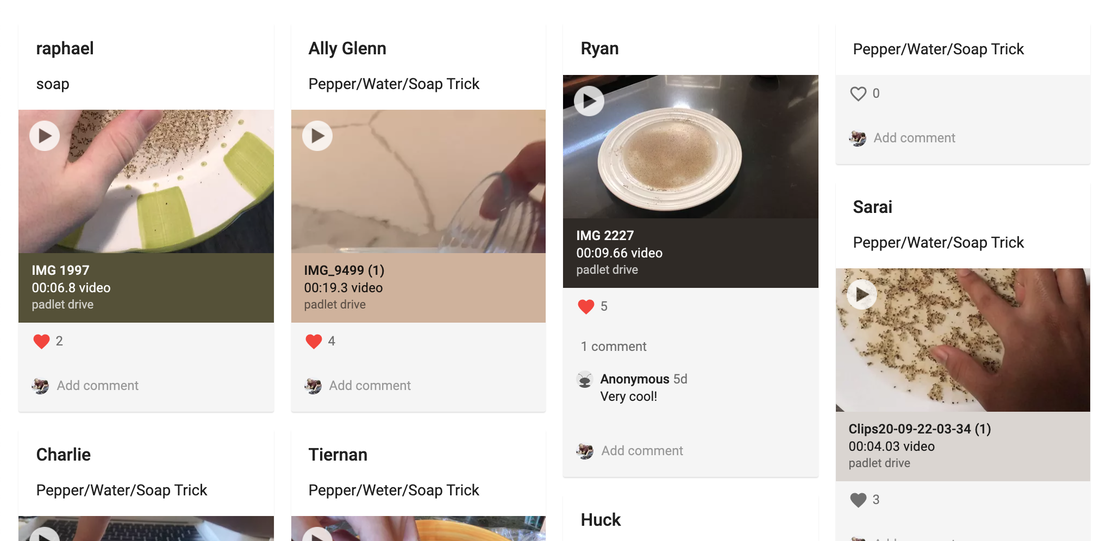
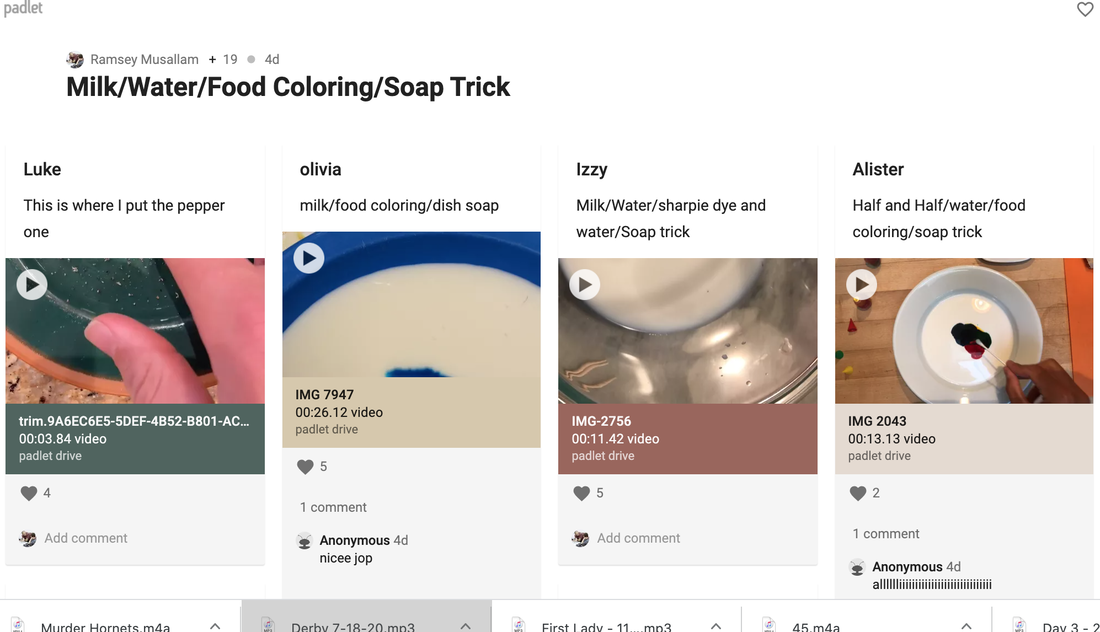
Continuing with my goal of bringing more SUSTAINABLE and ACCESSIBLE hands-on labs to the distance learning setting, below is another summary of the activity that was used to follow up the one described here. I hope you find it useful in your own planning for those of you that are still 100% distance. Step 1: Play this video for students to watch individually. Video is edited to strategically withhold the ending. Step 2: Challenge students to complete the experimental setup and post videos of conclusions on the Padlet board here. See screenshot of portion of Padlet board below. Students were given the below instructions: When you are ready, prop your camera over the dish (with help from a family member or holding with your other hand) record a video of you completing the experiment shown in the above video. Note: if you want to do it more than once, you need to rinse all soap out and start over. Step 3: Students viewed and commented on one another's work. Step 4: Students hypothesized, in Zoom breakouts: Why, on a molecular level, the soap-water combination forces the pepper to the side of the dish. Step 5: Students then repeated the above steps with this similar video edited to end prior to the experimental reveal. Video conclusions were posted here. See screenshot of portion of Padlet board below: Step 6: Students viewed this reveal video followed by a group discussion/lecture to fill in missing gaps and relate back to previous instruction.
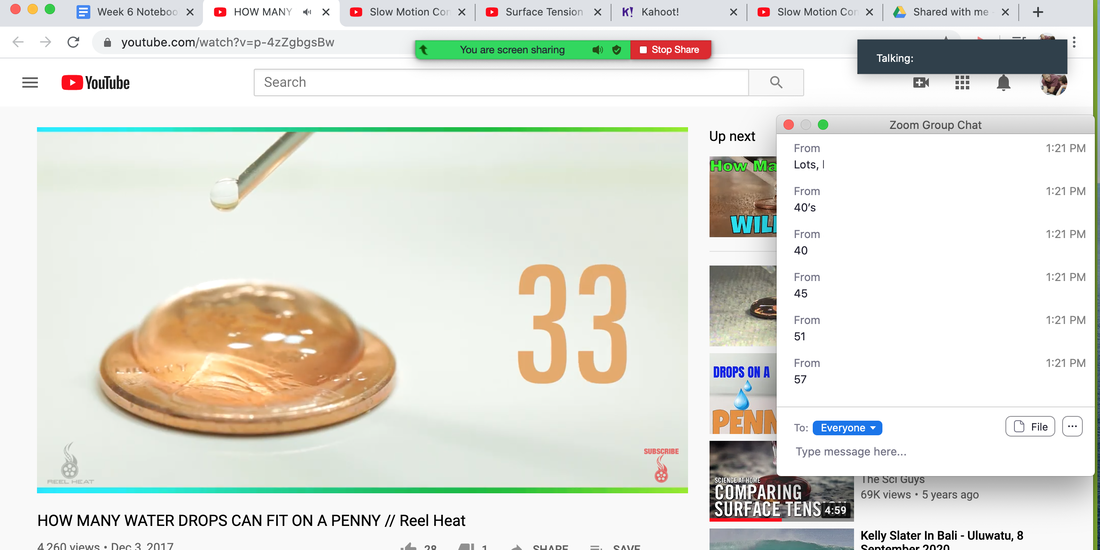
I wrote last about my desire to bring more hands-on learning to my 100% distance learning curriculum in chemistry and biology. I am going to begin this process SLOWLY leveraging materials students have at home, and as distance learning continues in our county, begin creating kits for students. I found this to be a fabulous resource for lab activities students can do at home (disclaimer: I realize I'm really behind on this and most STEM teachers are already doing fabulous stuff with students in the distance learning setting). Below is a summary of how I implemented my first SIMPLE activity. For pedagogical/content insight, students in this class just finished a unit on bonding and Lewis Structures. This week is a purely conceptual unit full of Intermolecular Forces activities (hence this lab). Once complete, we will circle back to Molecular Geometry and then build up an algorithmic understanding of polarity and IMFs. I am hoping the ability to circle back to this week as a reference point will provide a nice conceptual anchor. Anyways, below is the first guided inquiry activity. Click here for week's lesson document that students are working from. Step 1: Play this video and ask students to guess how many drops will fit on penny in the Zoom chat. See screenshot. Step 2: Challenge students to repeat experiment and post video of work here.
Step 3: Students viewed and commented on one another's work. Step 4: Students viewed this water and hypothesized, in Zoom breakouts, why you can fit more water on the penny than any other substance. Students were challenged to relate their hypothesis, in some way, back to the covalent Lewis structure for each substance (not: the goal of this portion was to create a clear sense cognitive tension and information gap awareness between what they know, Lewis Structures, and what they don't know, interaction BETWEEN lewis structure). Basically, I WANT them to mess this step up so that subsequent instruction has a cognitive anchor. Step 5: Students viewed this video to SLIGHTLY close the information gap in preparation for concrete instruction (see comments above) in the coming weeks. Essentially, the next few weeks are one larger Hero's Journey/5E Cycle. As distance learning pushes on, I keep revising and refining my list of tools. Because "tools" in many ways, represent our primary means of interacting with our students, I feel a professional obligation to always share. I'm sure this list will change in like 2 freaking days, but whatever...I'll share again :) l
1. Zoom
Distance Learning Lesson Planning Checklist (Catalyzed by Back to School Night reflection)9/18/2020
While many members of my PLN have gone back to teaching face-to-face via a myriad of different hybrid models, my colleagues and I still remain teaching in a 100% distance setting for the time being. Last night was "Back to School Night" via Zoom. The night was eye opening for me to say the least.
Normally Back to School Night is an energizing time for me connect with parents and share with them my authenticity as a teacher who values hands-on work and exploration with students in my science classes. Without realizing it, over the past ~ 6 months of distance learning, and clearly evident in my writing during this time, I have traded in my normal engaged, spontaneous, enthused presence in the classroom, for a stance that has placed protocols, organization, and structure, before all else. While tight structures are necessary, I hypothesize my fear of students feeling lost in the distance setting, and my lack of ability to work with them side-by-side, has forced me to see my teaching through a lens of structure first, rather than a lens of engagement and passion. If that makes sense? While I am sure many of us are experiencing similar trends in instructional behavior/design, this was underscored for me last night when interacting with parents via Zoom. While many appreciated the structure and my desire to organize their learning process, parents in my chemistry class specifically expressed a need for more hands on work at home, and engaged learning. This was a violating and dissonant thing to hear, as my normal stance as an educator is drenched in such a teaching framework. But how were they, specifically freshman parents, supposed to know that? I felt like it was Day 1, and all I had to show for my teaching was some boring Backwards Design Model, or tired lesson planning template that I was regurgitating from credential courses just 3 months prior. Indeed, in 100% distance learning, these are often the things that are on display. Such comments, while instructive, hit me hard, as hands on, engaged learning is something that comes so natural to me in the in the normal setting, something that parents/students rarely question...something that is the foundation of my authentic self in the classroom. If anything, the organization and systems I a putting in place have been weak spots for me as an educator over the years. Now they are front and center. After an evening of feeling sorry for myself, I woke up and jotted the below checklist to my normal teaching prep schedule in my "Notes" application. Each time I lesson plan moving forward, I plan on using this checklist to help me self regulate my lesson planning. A quality control measure to make sure I try my best (which will not always be feasible) to infuse my authentic self into the TIGHT STRUCTURE I have created for my distance learning. Rather than feeling guilty and trying to please parents...I simply need to do my best to infuse MYSELF into my lessons. "We teach from who we are" - Parker Palmer. Click here for my "Under Construction" Week 6 lesson plan/student notebook. If you look closely you will see opportunities for simple hands on work, collaboration synchronously and asynchronously, and more intentional sparking of student curiosity early on. This should be completely by end of day tomorrow, Sunday, 9/20. Although I'm sure this is not a new strategy for many of you, I am ABSOLUTELY loving the ability to not only hyperlink to specific portions within a Google Doc, but I am finding the ability to link to specific spots within a Google Doc from an EXTERNAL site very useful as well! So many applications...Click here for a quick tutorial if you don't know how to do this already. If you do, I hope distance learning is going well for everyone this Wednesday!
|
Categories
All
Archives
March 2024
|


 RSS Feed
RSS Feed