|
|
|
I LOVE finding video clips on YouTube and strategically editing them so that students are manipulated (for lack of a better word) into asking a content related question I want them to ask. If they ask it, they OWN it. Usually this process involves strategically trimming a video, perhaps removing the sound, covering up a portion of the video, or some combination of these techniques.
Although an invigorating process, the above protocol can be time consuming and intimidating for new teachers looking to curate video to spark student curiosity, without having to spend time bogged down in the details of various video editing software. Recently, I was struck with the realization that methods for sparking curiosity (see here and here for examples) can be done simply using the editing features in Google Slides.
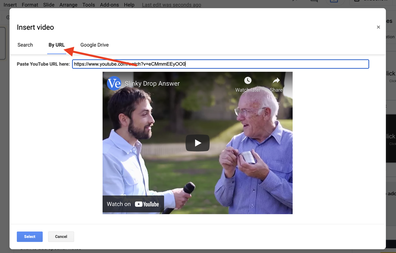
Step 1: Find a YouTube video that contains a clip, that when highlighted and edited strategically, will provoke questions about content. For example, the below video contains a clip of a slinky falling in slow motion to create a sense of inquiry around why the slinky appears to be suspended despite having been released from the individuals hand.
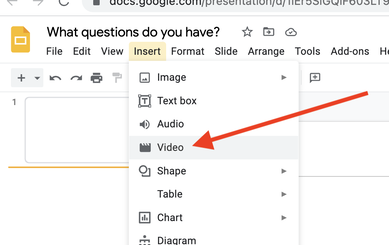
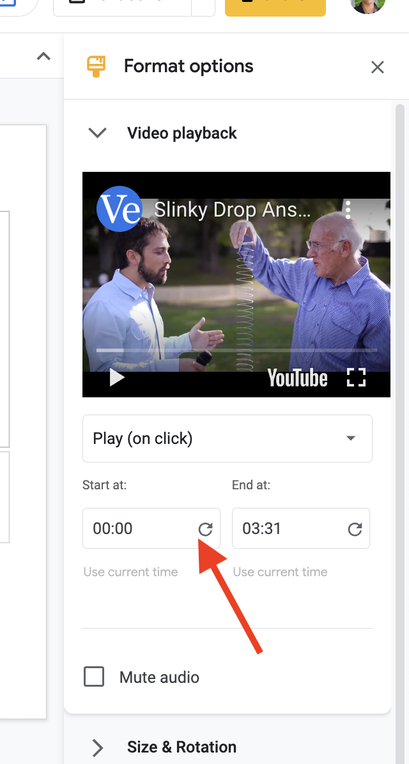
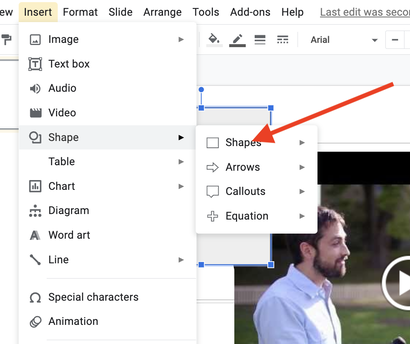
Step 2: Using the editing tools in Google Slides, insert and trim the video, as well as perform other editing functions such as removing audio, adding shapes to mask content, etc. to show the phenomena, but simultaneously create a riddle/Mystery Box effect. For example, the above video elicits more questions when the moment of drop is featured ONLY and the sound is removed entirely. See screenshots below.
Step 3: Play the edited video (in the slide) for students in presentation mode. Ask them "What are you curious about?". Alternatively, record a Screencast (Screencastify is a great free option), of the slide. This is particularly useful is attempting to spark student curiosity during online learning. See each option embedded below.
I recently discovered this amazing YouTube channel that proposes, WELL PRODUCED, video scenarios about perplexing "What if..." scenarios and then animates/describes what would occur? For example: "What if the oxygen disappeared from the world for 5 seconds?" or "What if the moon exploded?", etc. Keeping "What if" in mind, I have been struggling with hands-on labs during distance learning, and have recently been experimenting with leveraging the scenarios described in on the channel to empower "Thought Experiments" rather than forcing distance learning labs while student are not in class. I did two "What if" thought experiments this week, one in my Chemistry class, and one in my Biology class. Students LOVED THEM! The went like this:
Use "What if" thought experiments to spark curiosity about an upcoming unit (as I did above prior to a unit on Cellular Respiration), or challenge student's ability to apply learned content to a new, hypothetical situation. Either way, "What if" thought experiments have transformed engagement in my online classes this week.
I just finished the audiobook for the autobiography of the legendary Physics Professor Richard Feynman. A fabulous listen, I have always been inspired by Feynman's contagious curiosity and desire to learn all things with a seemingly vicious intensity. I find myself also drawn towards a similar, addictive need, to ask questions and search for answers to things that others might find meaningless or not directly relate to vocation. Or perhaps, I'm trying to justify my late night YouTube deep dives. Either way...Feynman's spirit speaks to me and listening to his life story added an unexpected sense direction to my own midlife contemplations. Through a lens of science instruction, I was endlessly inspired by the way Feynman's own personal sense of curiosity naturally mingled into his instructional pedagogy. That is, it was clear that he deeply values the question asking process and made it clear throughout the book that questions must precede any form of direct instruction or delivery of equations. My own passion for translating individual curiosity about the world into formal lesson plans was my motivation for this TED talk I delivered in 2013. This passion for "Delayed Direct Instruction" is intrinsic to the inquiry learning cycle and its efficacy in helping students develop conceptual models for understanding is outlined in the literature. Click here for example of the plethora of research articles that underscore the importance of questions BEFORE lecture. The first 30 seconds of this clip from The Challenger Disaster, a film about Feynman's role in uncovering the the true cause of the Space Shuttle Challenger disaster, clearly demonstrates Feynman's inability to separate conceptual understanding and curiosity from instructional pedagogy. I could watch this clip all day long! Perhaps the below quote sums up the purpose of this post and its relationship to Feynman best. I read the quote as Feynman challenging educators of today to find a way, despite the myriad forms of resistance, to figure out ways to spark student curiosity in search of authentic, conceptual understanding. I think, however, that there isn't any solution to this problem of education other than to realize that the best teaching can be done only when there is a direct individual relationship between a student and a good teacher - a situation in which the student discusses the ideas, thinks about the things, and talks about the things. It's impossible to learn very much by simply sitting in a lecture, or even by simply doing problems that are assigned. But in our modern times we have so many students to teach that we have to try to find some substitute for the ideal. - Richard Feynman I recently stumbled across this podcast between Tim Ferris and Seth Godin. About midway through the episode Tim begins to grill Seth about the concept of Pedagogy, and how he goes about thinking about learning and instruction through a lens of. cognition and motivation.
To quote Seth: "We learn things by becoming momentarily incompetent" This one quote summarizes so beautifully everything I tried to say here, and on my best days, what I try and do in the classroom (pandemic or not). That is, to carefully walk the line between the delivery, and the withholding of information. To have faith in the empty spaces. To honor the mystery box. I encourage you all to listen to Tim and Seth's thoughtful conversation. Click here to access the first of three recordings for my "Spark Learning" Masterclass. This session explored research and strategies related to sparking student curiosity at the beginning of an inquiry learning cycle.
|
Categories
All
Archives
March 2024
|

 RSS Feed
RSS Feed